Rivastigmine Hydrogen Tartrate
Breadcrumb
- Home
- Active Pharmaceutical Ingredient Products
- Rivastigmine Hydrogen Tartrate
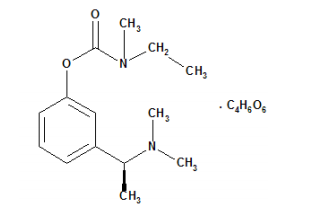
Rivastigmine Hydrogen Tartrate API
CAS Number: 129101-54-8
Breadcrumb
- Home
- Active Pharmaceutical Ingredient Products
- Rivastigmine Hydrogen Tartrate
About Rivastigmine Hydrogen Tartrate API
Therapeutic Category
Central Nervous System (CNS)
API Technology
Synthetic
Dose Form
Film (Transdermal), Oral Solid/Capsules
Dr Reddy's Development Status
Available
Available Regulatory Filing
USDMF, CEP Submitted, EUDMF, China DMF
Mechanism of Action
Rivastigmine is a type of medicine called an acetylcholinesterase inhibitor. It works by slowing the breakdown of a neurotransmitter in the brain called acetylcholine.
Indication
EXELON is an acetylcholinesterase inhibitor indicated for treatment of:
- Mild to moderate dementia of the Alzheimer’s type
- Mild to moderate dementia associated with Parkinson’s disease
Dr. Reddy's Expertise
Headquartered in Hyderabad, India, Dr. Reddy's Laboratories is one of the leading Active Pharmaceutical Ingredients (API) manufacturers and suppliers globally for Rivastigmine Hydrogen Tartrate API. Dr. Reddy's API business is a preferred partner to pharma companies across the US, Europe, Brazil, Latin America, Japan, China, Korea, Middle East and other emerging markets.
Dr. Reddy's API business thrives on the deep technical strengths established over the last 30+ years in the development and manufacture of complex APIs such as steroids, peptides, complex long chain molecules and highly potent APIs (HPAPIs / oncology drugs). This expertise is complemented by our prowess in intellectual property and regulatory affairs which helps us consistently meet and exceed regulatory standards. Dr. Reddy's Rivastigmine Hydrogen Tartrate API is the outcome of the extensive expertise in R&D, IP, and Regulatory.
A key component in helping our customers be first to market is a responsive supply chain. We achieve this by making sure that all our facilities are operating efficiently and to the latest standards of quality, safety, and productivity. A strong interconnect between business and factories allows for a quick reaction to dynamic market changes, so that we can avert shortages and meet sudden surges in demand.
Related Resources

How Indian API Pharma Companies Are Powering Global Drug Supply Chains
India has long been recognized as the “pharmacy of the world,” and its API pharma companies play a pivotal role in sustaining global drug supply chains. With a robust manufacturing ecosystem, skilled workforce, and regulatory expertise, API manufacturing companies in India are not only meeting domestic healthcare needs but also supplying critical active pharmaceutical ingredients to markets across North America, Europe, Asia, and Africa.
India’s Strategic Role in Global API Supply
India accounts for nearly 20% of global API exports, making it one of the largest suppliers of active pharmaceutical ingredients worldwide. The country’s API sector supports the production of generic drugs, branded formulations, and specialty medicines, enabling affordable healthcare access across continents.
Key strengths of API pharma companies in India include:
- Cost-effective production
- Strong regulatory compliance (US FDA, EMA, WHO GMP)
- Advanced process chemistry capabilities
- Scalable infrastructure for high-volume manufacturing
- API Manufacturing Companies in India: Global Reach and Capabilities
Leading API manufacturing companies in India such as Dr. Reddy’s Laboratories, Sun Pharma, Aurobindo Pharma, and Divi’s Laboratories have built world-class facilities and supply APIs for therapeutic areas including:
- Oncology
- Cardiovascular
- Anti-infectives
- Central Nervous System (CNS)
- Diabetes and metabolic disorders
These companies operate multiple manufacturing sites, many of which are approved by international regulatory agencies, ensuring uninterrupted global supply.
Handling Complex Chemistry and High Potency APIs
Indian API manufacturers are increasingly taking on complex chemistry projects, including:
- Multi-step synthesis of oncology APIs like Eribulin and Lenalidomide
- Peptide and steroid APIs requiring specialized containment
- Fermentation-based APIs for antibiotics and biologics
Their ability to handle high-potency APIs (HPAPIs) and niche molecules has positioned India as a preferred partner for global pharmaceutical innovators and generic drug makers.
Digitalization and Automation in API Pharma
To meet global quality standards and improve efficiency, API pharma companies in India are investing in:
- Automated reactors and process control systems
- Real-time quality monitoring using spectroscopic sensors
- Electronic batch records and data integrity platforms
- AI-driven predictive maintenance and process optimization
These technologies help reduce human error, improve scalability, and ensure consistent product quality across batches.
Resilience During Global Disruptions
During the COVID-19 pandemic and subsequent supply chain disruptions, Indian API manufacturers demonstrated remarkable resilience by:
- Rapidly scaling production of essential APIs like Paracetamol, Hydroxychloroquine, and Azithromycin
- Diversifying supply chains to reduce dependence on imports
- Collaborating with global partners to ensure continuity of drug supply
- This adaptability has reinforced India’s reputation as a reliable and strategic supplier in the global pharmaceutical ecosystem.
Government Support and Policy Initiatives
The Indian government has launched several initiatives to strengthen the API sector, including:
- Production Linked Incentive (PLI) Scheme for bulk drug manufacturing
- API parks and infrastructure development
- Support for R&D and technology upgrades
These policies aim to reduce import dependence, boost domestic capabilities, and enhance India’s competitiveness in global markets.
India’s API Export Strength: Key Statistics for 2025
India continues to be a dominant force in the global pharmaceutical supply chain, particularly in the API sector. Here are some compelling statistics that highlight the scale and impact of API pharma companies in India:
India is the 3rd largest API producer globally, holding an 8% share in the global API market
- Over 500 different APIs are manufactured in India, ranging from simple molecules to highly complex and potent compounds
- India supplies 57% of APIs listed on the WHO’s prequalified list, underscoring its role in global public health
- The Indian API industry is projected to grow at a CAGR of 13.7%, significantly outpacing the global generic API market
- In 2025, India’s pharmaceutical exports reached $50 billion, with APIs contributing a substantial portion of this figure
- Government initiatives like the Production Linked Incentive (PLI) Scheme have led to the setup of 261 new manufacturing sites, boosting domestic API capacity and export potential
These numbers reflect the strategic importance of API manufacturing companies in India in ensuring the availability of affordable, high-quality medicines worldwide.
Conclusion
API manufacturing companies in India are not just suppliers—they are strategic partners in global healthcare. With advanced capabilities, regulatory strength, and a commitment to innovation, API pharma companies in India are powering the world’s drug supply chains and shaping the future of affordable medicine.
 Read More
Read More

How Indian API Manufacturers Handle Complex Chemistry Projects
India has emerged as a global powerhouse in the active pharmaceutical ingredients (API) sector, with numerous API manufacturing companies in India leading the charge in innovation, scalability, and cost-efficiency. One of the most impressive capabilities of these companies is their ability to manage complex chemistry projects—from multi-step synthesis to handling hazardous reactions and developing niche molecules.
India’s Strength in API Manufacturing
The India API manufacturing company landscape is vast, with firms ranging from large-scale producers to specialized contract development and manufacturing organizations (CDMOs). These companies have built a reputation for:
- Robust process development capabilities
- Strong regulatory compliance
- Cost-effective production models
- Expertise in handling complex chemical reactions
- India supplies over 50% of APIs used in global generic drug production, and its manufacturers are increasingly taking on high-value, technically challenging projects.
Key Strategies for Managing Complex Chemistry
Advanced Process Development
Indian API manufacturing companies invest heavily in process R&D to optimize reaction pathways, reduce steps, and improve yields. Techniques include:
- Retrosynthetic analysis to simplify complex molecules
- Catalysis and biocatalysis for selective transformations
- Flow chemistry for safer and scalable continuous reactions
- These approaches allow companies to tackle APIs with multiple chiral centers, unstable intermediates, or low natural abundance.
Specialized Infrastructure and Equipment
Handling complex chemistry requires specialized infrastructure. Leading API manufacturing facilities in India are equipped with:
- High-pressure reactors for hydrogenation and nitration
- Cryogenic systems for temperature-sensitive reactions
- Isolated containment areas for potent or hazardous compounds
- This infrastructure enables safe and efficient execution of reactions that are otherwise difficult to scale.
Skilled Scientific Talent
India’s strength lies in its highly trained chemists and chemical engineers. Many professionals have global experience and advanced degrees, allowing them to:
- Troubleshoot complex synthesis challenges
- Develop scalable and reproducible processes
- Ensure compliance with international regulatory standards
- Companies often collaborate with academic institutions to stay at the forefront of chemical innovation.
Regulatory Expertise and Global Compliance
Indian API manufacturing companies are well-versed in regulatory frameworks such as:
- US FDA
- EMA (Europe)
- PMDA (Japan)
- WHO GMP
This expertise allows them to develop APIs for regulated markets, including oncology, antivirals, and CNS drugs, which often involve complex chemistry.
Use of Automation and Digital Tools
To manage complexity and ensure consistency, Indian manufacturers are increasingly adopting:
- Automated reaction monitoring systems
- Digital batch records and process analytics
- AI-driven predictive modeling for reaction outcomes
- These tools help reduce variability, improve safety, and accelerate development timelines.
Case Examples of Complex Chemistry Success
Several India API manufacturing companies have successfully developed APIs involving:
- Multi-step synthesis of anti-cancer drugs like Imatinib and Erlotinib
- Fermentation-based production of antibiotics such as Rifampicin
- Peptide synthesis for niche therapeutic areas
These projects demonstrate India’s capability to handle both synthetic and biologically derived APIs with precision and scalability.
Dr. Reddy’s Laboratories, a leading India API manufacturing company, has developed Eribulin as part of its oncology API portfolio. The synthesis of Eribulin is highly complex, involving:
- Multi-step synthesis with stereoselective reactions
- Handling of highly potent intermediates
- Stringent containment and purification protocols
Dr. Reddy’s has filed regulatory documentation such as USDMF, EUDMF, and Canada DMF, demonstrating its global compliance and technical capability.
Other Complex APIs by Dr. Reddy’s
Dr. Reddy’s is known for its expertise in developing and manufacturing highly complex APIs, including:
- Steroids: These require multi-step synthesis and precise control of stereochemistry.
- Peptides: Involve solid-phase synthesis and purification challenges.
- Oncology APIs: Such as Bortezomib, Lenalidomide, and Pomalidomide, which are highly potent and require specialized containment.
- Complex long-chain molecules: Used in antivirals and cardiovascular therapies.
- High Potency APIs (HPAPIs): Require dedicated facilities with advanced safety and containment systems.
The company’s ability to handle such complexity is supported by:
- State-of-the-art R&D centers
- Dedicated HPAPI manufacturing blocks
- Strong IP and regulatory teams
- Global supply chain integration
Why This Matters
The ability to manufacture complex APIs like Eribulin positions Dr. Reddy’s as a strategic partner for global pharmaceutical companies. It also highlights India’s growing role in the active pharmaceutical ingredients industry, especially in high-value, innovation-driven segments.
Conclusion
India’s API manufacturing facilities are not just cost-effective—they are centers of chemical innovation and technical excellence. By combining advanced infrastructure, skilled talent, and digital tools, API manufacturing companies in India are well-equipped to handle the most demanding chemistry projects. As global demand for complex APIs grows, India is poised to play an even more strategic role in the pharmaceutical supply chain.
 Read More
Read More

Key Design Considerations for Modern API Manufacturing Facilities
The active pharmaceutical ingredient industry is undergoing a transformative shift. As global demand for high-quality medicines continues to rise, manufacturers are embracing cutting-edge technologies to enhance efficiency, reduce environmental impact, and ensure consistent quality. This article explores how automation, artificial intelligence (AI), and sustainability are reshaping the future of API manufacturing.
Regulatory Compliance and GMP Standards
One of the foremost considerations in designing active pharmaceutical ingredients facilities is adherence to Good Manufacturing Practices (GMP). Regulatory bodies such as the FDA, EMA, and CDSCO mandate strict guidelines for facility layout, equipment, and operational protocols. Key design elements include:
- Cleanroom classifications to control contamination.
- Segregated zones for different stages of production.
- Validated HVAC systems to maintain air quality and pressure differentials.
- Compliance ensures product safety and facilitates global market access.
Facility Layout and Workflow Optimization
Efficient layout design is crucial for minimizing cross-contamination and optimizing workflow. Modern API manufacturing facilities often adopt:
- Linear or modular layouts to streamline material flow.
- Dedicated areas for synthesis, purification, drying, and packaging.
- Controlled access zones to restrict personnel movement and enhance biosecurity.
- A well-planned layout reduces bottlenecks and supports lean manufacturing principles.
Integration of Automation and Digital Systems
Automation is transforming the active pharmaceutical ingredients industry by improving consistency and reducing human error. Facility design must accommodate:
- Automated reactors and filtration systems.
- Real-time monitoring and control systems.
- Digital batch records and data integrity solutions.
- Smart facilities leverage IoT and AI to enhance productivity and ensure compliance with 21 CFR Part 11 and other data regulations.
Environmental Controls and Sustainability
Sustainability is a growing priority in API manufacturing facilities. Design considerations include:
- Energy-efficient HVAC and lighting systems.
- Solvent recovery units to reduce chemical waste.
- Water recycling and effluent treatment plants.
- Green design not only reduces environmental impact but also lowers operational costs and improves community relations.
Safety and Hazard Management
API production often involves hazardous chemicals and high-pressure reactions. Safety-focused design includes:
- Explosion-proof zones and equipment.
- Emergency ventilation and fire suppression systems.
- Safe storage for flammable and toxic materials.
- Risk assessments and HAZOP studies guide the placement of safety features and emergency protocols.
Scalability and Flexibility
Modern active pharmaceutical ingredients facilities must be adaptable to changing market demands. Design strategies include:
- Modular construction for easy expansion.
- Flexible production lines that can handle multiple APIs.
- Multi-purpose reactors and cleanrooms.
- Scalability ensures long-term viability and supports contract manufacturing opportunities.
Personnel and Training Infrastructure
Human factors are integral to facility performance. Design should support:
- Comfortable and hygienic staff areas.
- Training rooms and simulation labs.
- Clear signage and workflow instructions.
- Investing in personnel infrastructure enhances compliance and operational excellence.
Expanded Examples of Automation in API Manufacturing Facilities
Automation is a cornerstone of modern API manufacturing facilities, driving efficiency, consistency, and compliance. Here are several practical examples of how automation is being implemented across different stages of production:
Automated Reactors and Process Control Systems
- Programmable Logic Controllers (PLCs) and Distributed Control Systems (DCS) manage temperature, pressure, and mixing speeds in real-time.
- Automated dosing systems precisely add reagents and solvents, reducing variability and improving yield.
Robotic Material Handling
- Automated guided vehicles (AGVs) transport raw materials and finished APIs between zones, minimizing human contact and contamination risk.
- Robotic arms handle packaging, labeling, and palletizing tasks with high speed and accuracy.
Inline Quality Monitoring
- Spectroscopic sensors (e.g., NIR, Raman) integrated into production lines provide real-time analysis of chemical composition.
- Automated sampling systems collect and test product samples without interrupting the process.
Cleanroom Automation
- Airflow and pressure monitoring systems automatically adjust HVAC settings to maintain cleanroom classifications.
- Automated cleaning systems ensure consistent sanitation of equipment and surfaces, reducing downtime.
Digital Batch Record Systems
- Electronic Batch Records (EBRs) replace manual documentation, ensuring data integrity and compliance with 21 CFR Part 11.
- Audit trail automation tracks every change and action, simplifying regulatory inspections.
Predictive Maintenance
- IoT sensors monitor equipment health and performance.
- AI-driven analytics predict failures before they occur, allowing for proactive maintenance and reducing unplanned downtime.
Inventory and Supply Chain Automation
- Automated inventory tracking systems use RFID and barcode scanning to manage stock levels.
- Integrated ERP systems synchronize procurement, production, and distribution, improving supply chain visibility.
These automation technologies not only enhance operational efficiency but also support scalability, reduce human error, and ensure compliance with global regulatory standards.
Conclusion
Designing modern API manufacturing facilities is a multidisciplinary effort that balances regulatory requirements, technological innovation, and environmental responsibility. As the active pharmaceutical ingredients industry evolves, facility design will continue to play a pivotal role in ensuring safe, efficient, and sustainable drug production.
 Read More
Read More

The Future of API Manufacturing: Automation, AI, and Sustainability
The active pharmaceutical ingredient industry is undergoing a transformative shift. As global demand for high-quality medicines continues to rise, manufacturers are embracing cutting-edge technologies to enhance efficiency, reduce environmental impact, and ensure consistent quality. This article explores how automation, artificial intelligence (AI), and sustainability are reshaping the future of API manufacturing.
Automation in API Manufacturing
Automation is revolutionizing the way APIs are produced. Traditional batch processes are being replaced by continuous manufacturing systems, which offer several advantages:
- Improved Efficiency: Automated systems reduce manual intervention, speeding up production cycles.
- Enhanced Precision: Robotics and programmable logic controllers (PLCs) ensure consistent process parameters.
- Reduced Human Error: Automation minimizes variability and improves reproducibility.
- In the active pharmaceutical ingredient industry, automation also supports real-time monitoring and control, enabling manufacturers to detect deviations early and maintain product quality.
AI-Powered Process Optimization
Artificial Intelligence is becoming a game-changer in API manufacturing. AI algorithms analyze vast datasets from production lines to identify patterns, predict outcomes, and optimize processes. Key applications include:
- Predictive Maintenance: AI can forecast equipment failures, reducing downtime and maintenance costs.
- Process Modeling: Machine learning models simulate chemical reactions, helping chemists refine synthesis routes.
- Quality Control Enhancement: AI-driven image recognition and sensor data analysis improve defect detection and reduce waste.
- By integrating AI into manufacturing workflows, the active pharmaceutical ingredient industry is moving toward smarter, more agile production environments.
Sustainability in API Manufacturing
Environmental sustainability is no longer optional—it’s a strategic imperative. The API manufacturing process often involves hazardous chemicals and energy-intensive operations. To address these challenges, companies are adopting:
- Green Chemistry Principles: Designing chemical processes that minimize waste and use safer reagents.
- Solvent Recovery Systems: Recycling solvents to reduce environmental impact and operational costs.
- Energy-Efficient Equipment: Implementing technologies that lower energy consumption and carbon footprint.
Sustainability initiatives not only align with global environmental goals but also enhance brand reputation and regulatory compliance in the active pharmaceutical ingredient industry.
Regulatory and Industry Implications
As automation, AI, and sustainability become integral to API manufacturing, regulatory bodies are updating guidelines to reflect these advancements. Manufacturers must ensure that new technologies comply with Good Manufacturing Practices (GMP) and data integrity standards. Industry collaborations and public-private partnerships are also fostering innovation, with shared platforms for AI development and green technology adoption.
Continuous Manufacturing: The Future Standard
Traditional batch processing is being replaced by continuous manufacturing, which offers:
- Higher efficiency and reduced production costs.
- Improved scalability and faster time-to-market.
- Enhanced product consistency through real-time quality monitoring.
- This shift is supported by both automation and AI, creating a seamless, data-driven production environment.
Key AI Impact Statistics in API and Pharma Manufacturing
Economic Value:
AI is projected to generate between $350 billion and $410 billion annually for the pharmaceutical industry by the end of 2025. This value comes from improvements in drug development, clinical trials, precision medicine, and commercial operations.
Adoption Rate:
- 80% of pharmaceutical professionals now use AI in drug discovery workflows
- 95% of pharma companies are actively investing in AI capabilities
Efficiency Gains:
- AI has been shown to reduce clinical trial costs by up to 70%.
- Development timelines have been cut by as much as 80%, significantly accelerating time-to-market
Market Growth:
The global AI in pharmaceuticals market is valued at $1.94 billion in 2025 and is expected to grow at a CAGR of 27%, reaching $16.49 billion by 2034
- AI has been shown to reduce clinical trial costs by up to 70%.
- Development timelines have been cut by as much as 80%, significantly accelerating time-to-market
Specialized Applications:
- AI in cancer diagnostics is growing at a CAGR of 40.1%.
- AI in genomics, crucial for precision therapeutics, is expanding at 52.7% CAGR.
These statistics highlight how AI is not just enhancing R&D but also transforming API manufacturing by optimizing synthesis routes, improving quality control, and enabling predictive maintenance.
Conclusion
The future of the active pharmaceutical ingredient industry lies in embracing innovation. Automation streamlines operations, AI unlocks new efficiencies, and sustainability ensures long-term viability. Together, these pillars are shaping a smarter, cleaner, and more resilient API manufacturing landscape
 Read More
Read More

End-to-End Overview of Pharma API Manufacturing
In the pharmaceutical industry, the term API pharma refers to the Active Pharmaceutical Ingredient—the biologically active component in a drug product. The process of API manufacturing is a complex, highly regulated, and critical part of drug development. This article provides a comprehensive overview of the end-to-end journey of API manufacturing, from raw material sourcing to final quality control.
What is an API in Pharma?
An API is the substance in a pharmaceutical drug that produces the intended effects. For example, in a pain relief tablet, the API is the compound that alleviates pain. Other components in the drug, known as excipients, help deliver the API effectively but do not have therapeutic effects themselves.
Stages of API Manufacturing
Raw Material Procurement
The first step in API manufacturing involves sourcing high-quality raw materials. These materials can be chemical compounds, biological substances, or plant-based extracts. The quality and consistency of these inputs are crucial, as they directly impact the efficacy and safety of the final product.
Chemical Synthesis or Fermentation
Depending on the nature of the API, manufacturers use either chemical synthesis or biotechnological methods like fermentation. Chemical synthesis involves multiple reaction steps to build the desired molecule, while fermentation uses microorganisms to produce complex biological APIs.
Purification and Isolation
After synthesis, the API must be purified to remove impurities and by-products. Techniques such as crystallization, filtration, and chromatography are commonly used. This step ensures that the API meets stringent purity standards set by regulatory authorities.
Drying and Milling
Once purified, the API is dried and milled into a fine powder. This step is essential for achieving the correct particle size, which affects the drug’s solubility and bioavailability.
Quality Control and Testing
Quality control is a cornerstone of API pharma manufacturing. Each batch undergoes rigorous testing for identity, potency, purity, and stability. Analytical methods like HPLC (High-Performance Liquid Chromatography) and mass spectrometry are employed to ensure compliance with pharmacopeial standards.
Analytical Testing
Analytical testing is the backbone of QC in API pharma. It involves:
- Identity Testing: Confirms the chemical structure of the API using techniques like Nuclear Magnetic Resonance (NMR) and Infrared Spectroscopy (IR).
- Purity Analysis: Determines the level of impurities using High-Performance Liquid Chromatography (HPLC), Gas Chromatography (GC), and Mass Spectrometry (MS).
- Potency Testing: Measures the concentration of the API to ensure it matches the labelled strength.
- Residual Solvent Testing: Ensures solvents used during synthesis are within acceptable limits.
Microbiological Testing
For APIs that are biologically derived or used in sterile formulations, microbiological testing is essential. This includes:
- Bioburden Testing: Assesses the microbial load in the API.
- Endotoxin Testing: Detects pyrogens that can cause fever if injected.
- Sterility Testing: Ensures the absence of viable microorganisms in sterile APIs.
Stability Studies
Stability testing evaluates how the API behaves under various environmental conditions over time. These studies help determine:
- Shelf Life
- Storage Conditions
- Packaging Requirements
- Accelerated and long-term stability studies are conducted as per ICH guidelines to simulate real-world scenarios.
Physical Property Testing
Physical characteristics like particle size, polymorphism, and hygroscopicity can affect the API’s performance. Techniques such as X-ray Powder Diffraction (XRPD) and Differential Scanning Calorimetry (DSC) are used to analyze these properties.
Documentation and Traceability
Every QC test is meticulously documented to ensure traceability and compliance. Batch records, Certificates of Analysis (CoA), and audit trails are maintained to satisfy regulatory inspections and customer audits.
Regulatory Compliance Audits
QC teams also prepare for periodic audits by regulatory bodies such as the FDA, EMA, and CDSCO. These audits assess the robustness of the QC systems, data integrity, and adherence to Good Manufacturing Practices (GMP).
Regulatory Compliance in API Manufacturing
API manufacturers must adhere to Good Manufacturing Practices (GMP) and comply with regulations from bodies like the FDA (U.S.), EMA (Europe), and CDSCO (India). These regulations cover everything from facility hygiene to documentation and traceability.
Challenges in API Pharma Manufacturing
Supply Chain Disruptions: Global events can impact the availability of raw materials.
Environmental Regulations: Chemical synthesis often involves hazardous substances, requiring strict environmental controls.
Cost Pressures: Balancing quality with cost-efficiency is a constant challenge.
Future Trends in API Manufacturing
The industry is witnessing a shift toward green chemistry, continuous manufacturing, and AI-driven process optimization. These innovations aim to make API pharma production more sustainable, efficient, and responsive to market needs.
Conclusion
The journey of API manufacturing is intricate and vital to the pharmaceutical supply chain. From raw material sourcing to final quality checks, each step is governed by precision, regulation, and innovation. As the demand for high-quality medicines grows, the role of robust API manufacturing processes becomes increasingly important.
 Read More
Read More

The Impact of Global Regulations on the Active Pharmaceutical Ingredient Industry
The Active Pharmaceutical Ingredient (API) industry is a cornerstone of the global pharmaceutical sector, responsible for producing the essential components that give medications their therapeutic effects. As the industry continues to evolve, it faces a complex landscape of global regulations that significantly impact its operations, growth, and innovation.
Ensuring Quality and Safety
One of the primary objectives of global regulations is to ensure the quality and safety of APIs. Regulatory bodies such as the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and the International Council for Harmonisation (ICH) set stringent guidelines that manufacturers must adhere to. These regulations cover every aspect of API production, from raw material sourcing to manufacturing processes and quality control. Compliance with these standards is crucial to prevent contamination, ensure consistency, and guarantee that APIs meet the required safety and efficacy standards.
Navigating Regulatory Challenges
The API industry must navigate a myriad of regulatory challenges that vary by region. For instance, the FDA's Current Good Manufacturing Practice (CGMP) guidelines are particularly rigorous, requiring manufacturers to maintain high standards of cleanliness, employee training, and documentation2. Similarly, the EMA's guidelines emphasize the importance of scientific evaluation and safety monitoring. These regulations can pose significant challenges for manufacturers, especially those operating in multiple regions, as they must stay updated with the latest regulatory changes and ensure compliance across different jurisdictions.
Impact on Innovation and Market Access
While regulations are essential for maintaining high standards, they can also impact innovation and market access. The process of obtaining regulatory approval for new APIs can be lengthy and costly, potentially delaying the introduction of innovative therapies to the market. Additionally, smaller manufacturers may struggle to meet the stringent requirements, limiting their ability to compete with larger, more established companies. However, these regulations also drive the industry towards higher standards of quality and safety, ultimately benefiting patients worldwide.
Global Supply Chain and Manufacturing
The globalization of the API supply chain has introduced both opportunities and challenges. On one hand, it has enabled manufacturers to source raw materials and produce APIs at lower costs. On the other hand, it has increased the complexity of the supply chain, making it more vulnerable to disruptions. Regulatory measures aimed at ensuring the resilience and transparency of the supply chain are crucial in mitigating these risks. For example, the concentration of API manufacturing in specific regions, such as India and China, has raised concerns about supply chain vulnerabilities. Promoting geographic diversity in manufacturing can help reduce these risks and ensure a stable supply of APIs.
Conclusion
Dr. Reddy's Laboratories addresses these challenges by maintaining stringent compliance with global regulatory standards and investing in robust quality control systems. The company leverages strategic partnerships and collaborations to navigate diverse regulatory landscapes and ensure the consistent quality of its APIs. By focusing on innovation and maintaining a resilient supply chain, Dr. Reddy's continues to deliver high-quality APIs, meeting global market demands and enhancing patient care worldwide.
 Read More
Read More

The Importance of Supply Chain Management and Dr. Reddy’s Excellence
In today’s fast-paced and highly competitive market, effective supply chain management is crucial for the success of any organization. It ensures that products are delivered on time, inventory levels are optimized, and customer satisfaction is maximized. Dr. Reddy’s Laboratories has demonstrated exceptional prowess in this area, achieving significant milestones that set them apart in the industry.
Key Achievements:
Enhanced OTIF (On time in full) Delivery
Dr. Reddy’s commitment to delivering products on time and in full (OTIF) has seen remarkable improvement. This dedication ensures that customers receive their orders exactly when and how they need them, fostering trust and reliability with partners. The enhancement in OTIF delivery has been pivotal in building strong, long-lasting relationships with customers and stakeholders.
Reduced Back Orders
By streamlining supply chain processes, Dr. Reddy’s has successfully minimized back orders. This achievement not only ensures a more efficient and responsive supply chain but also leads to increased customer satisfaction and operational efficiency. The reduction in back orders means that customers can rely on Dr. Reddy’s to meet their needs promptly, enhancing the overall customer experience.
Integrated Demand and Supply Chain Management
Dr. Reddy’s innovative approach to integrating demand and supply chain management has been a game-changer. This integration allows the company to better anticipate customer needs, optimize inventory levels, and respond swiftly to market changes. By aligning demand with supply, Dr. Reddy’s can ensure that the right products are available at the right time, reducing waste and improving service levels.
Best Practices in Supply Chain Management
Dr. Reddy’s follows several best practices in supply chain management:
Leveraging Technology
Dr. Reddy’s utilizes advanced technologies such as AI and machine learning to forecast demand accurately and manage inventory efficiently. Real-time data analytics help in making informed decisions, reducing lead times, and improving overall supply chain visibility.
Supplier Relationship Management
Building strong relationships with suppliers is crucial. Dr. Reddy’s collaborates closely with the suppliers to ensure quality, reliability, and timely delivery of raw materials. This partnership approach helps in mitigating risks and enhancing supply chain resilience.
Continuous Improvement and Lean Practices
Adopting lean practices and a culture of continuous improvement allows Dr. Reddy’s to eliminate waste, streamline processes, and enhance productivity. Regular audits and feedback loops ensure that the supply chain remains efficient and responsive to changes.
Sustainability Initiatives
Dr. Reddy’s is committed to sustainability in the supply chain operations. We focus on reducing the carbon footprint, optimizing resource usage, and implementing eco-friendly practices. This commitment not only benefits the environment but also aligns with the growing demand for sustainable business practices.
Talent Development
Investing in the development of supply chain professionals is a priority for Dr. Reddy’s. We provide ongoing training and career development opportunities to the employees, ensuring that we have the skills and knowledge to manage complex supply chain operations effectively.
Conclusion
Dr. Reddy’s Laboratories’ achievements in supply chain management highlight the importance of a well-coordinated and efficient supply chain. Our commitment to enhanced OTIF delivery, reduced back orders, and integrated demand and supply chain management sets us apart in the industry but also ensures that we continue to meet and exceed customer expectations.
By following best practices such as leveraging technology, managing supplier relationships, continuous improvement, sustainability initiatives, and talent development, Dr. Reddy’s remains a trusted and reliable partner in the pharmaceutical industry, delivering excellence through their superior supply chain practices.
 Read More
Read More

Tentative Approval for Generic Lumateperone capsules

In a remarkable stride for innovation and speed, Dr. Reddy’s Laboratories has received Tentative Approval for Generic Lumateperone capsules a milestone that marks a new benchmark in small molecule development. Filed on NCE-1, this submission reflects our commitment to strategic excellence and scientific precision. Lumateperone completed DMF adequacy and received Tentative Approval in just 20 months, one of the fastest timelines in recent years for a small molecule. This is more than just a regulatory milestone. It’s a testament to our agility, collaboration, and relentless pursuit of quality. Congratulations to everyone who made this possible. For queries contact us at [email protected]
 Read More
Read More
Related APIs
Disclaimer
No information in this catalog - including any reference to any product or service - constitutes an offer for sale, or be construed as representing an offer for sale. Products protected under valid patents are not offered or supplied for commercial use. However, the research quantities of such products may be offered for the purpose of regulatory submissions, wherever such regulatory exemptions exist. The buyers should make their independent evaluation of the patent scenario for their respective markets and will be responsible for all patent related liabilities. Products protected under valid patents in India are not available for commercial use but would be available for Section 107A purposes.
Insights Delivered
Sign-up for our email service to get Market and Product insights and updates right to your digital doorstep
The categories of personal information collected in this form include name, company, and contact information etc. The personal information collected will be used for exploratory discussions on contract manufacturing, marketing and to perform research and analytics and others. For more information about the categories of personal information collected by Dr.Reddy's and the purposes for which Dr.Reddy's uses personal information, visit https://api.drreddys.com/privacy-policy.
Disclaimer
No information on this website, including any reference to any product or service constitutes an offer for sale or be construed as representing an offer for sale. Products protected under valid patents are not offered or supplied for commercial use. However, in certain cases, at Dr. Reddy's sole discretion, and subject to local legal requirement, the research quantities of such products may be offered for the purpose of regulatory submissions under Section 107A of the Indian Patent Act (Bolar exemption), wherever such regulatory exemptions exist. The buyers should make their independent evaluation of the product or service including, patent scenario in their respective markets and will be responsible for all patent related liabilities Dr. Reddy's disclaims all warranties, express or implied, including but not limited to warranties of merchantability, fitness for a particular purpose and non-infringement.


